Research Article - (2020) Volume 8, Issue 3
Studies on the ethnobotany and phytochemical on three species of wild Zingiber (Zingiberaceae) namely Z.Z. pseudopungens, Zingiber chlorobracteatum and Zingiber coloratum were conducted in Timbou Village, Tambunan District, Sabah, Borneo, Malaysia, Alab Mount and Mahua waterfall, Crocker Range from 1st Oct. until 7th Oct 2019. The objectives of these studies were to survey the ethnobotanical uses of the three wild Zingiber species used by the local Dusun communities of Tambunan, to extract essential oils from the major Zingiber species and lastly to determine the chemical components in the essential oils and to test it against bacteria. Ethnobotanical uses for these three Zingiber species are as follow, Z. pseudopungens leaves and stem are bitter; Z. viridifalvum sap is drinkable, medicine as anti-venom for snake-bite, pith cooked as soup and whole plant is for decorations and Z. coloratum plant is used as decorations, rhizome boiled made into soup and medicinal to treat diabetes. In this study, the preliminary phytochemical screening in both polar extract of ethanol and non-polar extract of n-hexane showed the presence of major secondary metabolites in these 3 types of studied Zingiber spp. GC-MS profiling gave the composition of few major compounds. Moreover, antibacterial assays of these oils had demonstrated that the Gram-positive bacteria in this study were more sensitive to the Zingiber spp. samples compared to the Gram-negative bacteria.
Ethnobotany; Phytochemical; Zingiber; Zingiberaceae
Plants extracts gain much of attention from a lot of communities especially scientists from all over the world. Malaysia’s tropical rainforests have vast amount of plants species which may produce essential chemical contents. It is estimated about 12,000 plants species in Malaysia and more than 1,000 species are said to have therapeutic properties and applied in the locals ’ traditional medicine [1]. Modern trends in medicine, research and consumer preference have increased the demand for natural products from plants [2]. Thus, plants have gained prominence due to theirs long term use by societies in traditional healthcare application. Numerous number of plants have been recognized by societies for their antifungal, antimicrobial, insecticidal, cytostatic and other therapeutic properties [3].
Few years ago, research in the field of natural products chemistry has improved exponentially due to development of analytical equipment and easy access to scientific information. This field leads to more implementation on drug discoveries from natural products. 23 new drugs derived from natural products were recognized between 2001 and 2005 for the treatment of disorders such as bacterial and fungal infections, cancer, diabetes, atopic dermatitis, Alzheimer ’ s disease and genetic diseases such as tyrosinaemia and Gaucher disease [4]. Besides that, development of chemical screening protocol for natural products has improved the production of natural products, methodologies of organic chemistry production, combination of biosynthesis processes and microbial genomics which are the several examples of recent technological innovations of new methods that fasten the work on drug discoveries [4].
Essential oils and extracts obtained from many plants have been utilized for various purposes such as for food, medicine, and cosmetics. An estimate of 3,000 essential oils are known and more than 3,000 have been found important in commercial market as flavours and fragrances. Essential oils are aromatic liquids which are obtained from a variety of plant parts such as flowers, rhizomes, leaves, piths, buds, seeds, barks and fruits. The oils of plants may be obtained by expression, fermentation or extraction. Steam distillation and hydro distillation are commonly used for extraction and commercial production of essential oils.
A lot of ethnobotanical studies on Zingiberaceae family have been initiated in North East India. Zingiberaceae is also common as medicinal plant in Thailand and Indonesia. However, not much information is available on the utilization of the members of this family in Sabah besides the commonly used Z. officinale. The natives of Sabah particularly the Kadazan/Dusun people are known to utilize plants around their surroundings as natural resources and as a source of herbal medicines, traditional food and raw materials for uncountable of domestic uses. Besides that, documentation of the medicinal herbs by ethnics in Sabah has been extensively studied especially by the Kadazan/Dusun community. Although many have done the traditional knowledge documentation, but the phytochemical study of the wild Zingiber species is still lacking. Therefore, this research will endeavour to extract the essential oil and determine their composition using Gas Chromatography-Mass spectrometry (GC-MS).
The Objectives of our studies are, namely to survey the ethnobotanical uses of wild Zingiber species by the local community of Tambunan, to collect specimens used traditional and to document their morphological features (Voucher specimens will be prepared), to extract essential oil from the major Zingiber species used in traditional medicine in Tambunan and to determine the chemical components in the essential oil and to test it against bacteria.
Tropical rainforest approximately cover seven percentage of earth’s surface which is around 12 million km2 and this forest type contains about 75% of the total known species on earth. Tropical rainforest is highly diverse with up to 473 tree and liana species coexisting in a single hectare. There are a few of density dependent effects which play the role as biotic factors on seed and seedling survivorship essential in maintaining biodiversity especially the trees in this forest type. Different types of niche among trees species with the availability of light and soil resources also contribute to the variety of plant diversity in this forest [5].
Tropical rainforest is categorized into several layers. Well known layer is canopy, which is around 30-40 m high. The upper levels of the rainforest receive more rain, wind and sunlight sources. Whereas, the biodiversity at the lower levels are stiller, shadowed and humid. Thus, most of the plants at canopy level act as a giant umbrellas, which may only allow two percentage of sunlight to reach the forest floor. Hence, rainforest floor often clean naturally and beneficial for a thick mat of interconnecting roots, climbing vines and epiphytes grow on, up, around and through the tree growths. Therefore, the structure of tropical rainforest leaves are often found thicker, tough and glossy [6].
Generally, tropical rainforest is rich in the biodiversity. Around 50% of living organisms are available within it. According to NAS, there are approximately 1,500 species of flowering plants and 750 species of trees over 1,000 hectare patch of rainforest. In Borneo, there are around 2,500 native tree species which are almost twice larger than the native tree species in United Kingdom [6]. Thus, most of Zingiber spp. are found in the tropical rainforest. They tend to be shrub-like. Gingers distributed widely in rainforest due to good quality of soil which is suitable for their growth. Also, rainforest’s climate is suitable for gingers growth because they prefer part-day shade and warm climates.
The term ethnobotany was first used by Harshberger in 1896. His definition of ethnobotany is the study of plants used by primitive and local community in certain places. Many scholars in the area are then redefined and reinterpreted the ethnobotany. Robbins, Harrington and Freire-Marreco broadened the definition of ethnobotany in 1916 where they expressed his idea that the science of this field should comprise the study and evaluation of the knowledge of all life cycle amongst primitive societies and of the implications of the vegetal surrounding upon the life customs, beliefs and history of these tribal peoples. Twenty-five years after ethnobotany was redefined, Jones advanced better definition where ethnobotany term is defined as the learning of interrelationships of primitive men and plants. In 1976, Schultes expanded this to include the relationships between man and his environmental surrounding vegetation. Yet, the broadest definition of ethnobotany is defined by Martin in 1995, where this term is described as the subdiscipline of ethnoecology that is concerned with local people’s interaction with plants.
Ethnobotany is a multidisciplinary endeavour that is usually categorized into 4 elements, which are; basic documentation of traditional botanical knowledge, quantitative evaluation of the use and management of botanical resources, experimental assessment of the benefits derived from plants and lastly implemented projects to increase the value that local people attain. There are a few of issues saying ethnobotanical studies should go beyond collecting, identifying, recording and producing a list of species taxonomy which may include their traditional usage. Ethnobotany will become more crucial field when its study has progressed to the point where results can be studied comparatively and has valid reason behind the scientific study. Thus, generally ethnobotany is the scientific study of plants importance in local culture for consumption, medicine, rituals, building, household implements, entertainment equipment, shelter and other purposes [7].
The interaction between plants with a variety of ethnics in Malaysia is common knowledge among Malaysians but the detail and scientific studies on them is lacking. The natives of the Malay Peninsula, Sabah and Sarawak had used plant and plant materials for a variety of uses in their daily lives. Unfortunately, ethnobotany has not been studied and research until now specifically in Tambunan District. Plants play major roles in the life of all Malaysian communities as they have been used as medicines, poisons, clothing, dyes, foods, drinks, building materials and many more [8].
In Sabah, there are about 50 ethnics. They have provided an essential heritage on the knowledge of the uses of plants in traditional healthcare and utilize the diversity of plants as food and traditional medicine in their daily basis. While, in Sarawak, the study on the traditional medicines has been growing exponentially since the mid-seventies. The plants used comprise trees, shrubs, herbs, epiphytes and climbers. Plants application are widely used among the locals of Sarawak which include external ailments such as pain, diarrhea, blood in stool, cough, asthma and many more. Also, there is a tribe of local people in Malaysia known as Temuan which comes from the Orang Asli community. They are well recognized by their belief of incorporating plants in ritual practices which may involve spirits or without association of spirits.
Plants belonging to Zingiberaceae family are commonly known as ginger and it is one of the major medicinal herbs which have strong aromatic and properties. Variety of Zingiberaceae species are indigenous to tropics particularly South Asia and Southeast Asia represent by almost 50 genera and 1300 species [9]. The species are commonly distributed in India, tropical Asian and extended to northern Australia [10]. Zingiberaceae family is well known throughout the world since the emergence of mankind because of its multipurpose uses such as in culinary and as medicinal herbs.
The genus Zingiber consists about 85 species of herbs mostly found in East Asia and tropical Australia. Availability of bioactive compounds such as gingerols, which are antibacterial agents and shogaols [11], diarylheptanoids [12], phenylbutenoids [13], flavanoids [14], diterpenoids [15], and sesquiterpenoids have been revealed through phytochemical studies on the several rhizomes of Zingiber spp.
Zingiberaceae family has two subfamilies which are Costideae and Zingiberoideae. Zingiberaceae is a plant family of flowering plants consisting of aromatic perennial herbs with tuberous rhizomes. Large herbaceous plants with distichous leaves with basal sheaths overlap to form a pseudostem. Zingiberaceae is an epiphytic plant. Their flowers are zygomorphic determinate cymose inflorescences and subtended by conspicuous, spirally arranged bracts. Zingiberaceae perianth is composed of two whorls, a fused tubular calyx, and a tubular corolla with one lobe larger than the other two. Flowers typically have two of their stamenoids fused to form a petaloid lip and have only two nectaries, the stigma is funnel-shaped [16].
Ginger is sold commercially as a fresh, as preserved ginger, as a pungent extract oleoresin and as steam distilled essential oil [10]. It was found that Zingiberaceous plants are part of economic importance and daily human needs to the community as a lot of the species are used as vegetables, spices, and condiments. It is also edible fruits, dyes, ornamentals and can be used as medicinal plants in the treatment of different diseases and ailments such as asthma, bronchitis, diarrhea, cough, cold and inflammation.
In Malaysia, Zingiber officinale and Zingiber cassumunar are used to treat asthma and rheumatism disease. In addition, Zingiber officinale has been widely used in traditional Chinese medicine to treat a wide range of ailments such as common cold, pain, digestive problems, and gastrointestinal disorders [17].
This family of ginger shows its valuable contribution to the people's livelihood as there are indications of its decreasing in population due to the high demand for consumption, utilization, deforestation, and exploitation. Thus, interventions on its conservations are required. Interventions of the ginger family may include in-situ and ex-situ modes of conservation, identification of all vital valuable Zingiberaceae plants and domestication of these plants [18].
Phytochemical studies
Since the early of 20th century, the characterization of organic chemistry structures has been done with different physiochemical methods. Innovations advances in terms of mass-spectrometry both in chromatographic techniques and identification tools enable us to face all the possibilities of separation and characterization of the increasing complexity compounds of plants [19].
Phytochemical derives from the Greek word “Phyto” for plant. It can also refer to the entire chemical constituents present naturally in plants, which have the ability to perform the function either by itself or combine with vitamins and others nutrients in food to prevent, stop and reduce the implication of diseases. Thousands of known phytochemicals such as carotenoids, flavonoids, steroids, tannins, glycosides, alkaloids, saponins, and many more [20]. Moreover, World Health Organization (WHO) has defined several of plants species with those chemical properties or compounds are crucial in therapeutic purposes [21].
Phytochemical study in Malaysia was first started in June of year 1952 by Arthur. There were around 200 types of plant species collected from Kota Kinabalu (Jesselton), Ranau and Keningau. All the samples were screened for their chemical contents such as alkaloids, triterpenoids, and steroid compounds, saponins, essentials oils, glucosides and, hydrogen cyanide [22]. Although with a great initial of phytochemical study in Sabah by Arthur (1952), there was a long period of inactivity until University Kebangsaan Malaysia cooperated with Sabah Parks to carry out an expedition in Danum-Valley in year 1987. Few years later, Ahmad in Gunung Danum Valley in 1989 and Tawau Hill in 1993 continued the phytochemical study. The phytochemical survey in Sayap was another effort from UKM and Sabah Parks to determine the potential uses of plants as a source of useful valuable chemicals and pharmaceutical products. Nowadays, the phytochemical studies fuel the efforts toward the discovery of useful drugs from the natural resources in the tropical rainforest.
The advent of allopathic medicine, using plants for medicinal purposes is still prevalent in many parts of the world yet medicinal plants have been used for the treatment of diverse ailments for thousands of years. For instance, Zingiber spp. such as Zingiber cassumunar has been applied in Thai traditional medicine for treating muscle pain and inflammation. The pulverized dried rhizomes are also given orally as an antiasthmatic in Thailand. Ethanolic extract from the rhizomes was found to have anti-spasmodic activity. Oral administration of capsules containing pulverized dried rhizomes to asthmatic children showed a positive effect in relieving asthmatic symptoms with low side effects [10].
Essential oils are compounds extracted from aromatic plants, and some essential oils are considered as effective inhibitors of food-borne pathogens and generally used in food preservation. Essential oils also known as aromatic oils, fragrant oil, ethereal oil and steam-volatile oil can be extracted from different part of aromatic plants such as flowers, leaves, stems, roots, seeds, bark, resin or fruit. Essential oils extracted by hydrodistillation, steam distillation and organic solvent extraction are basically contain volatile and highly concentrated substances.
According to international definition, essential oil is distillation products that are not soluble in water and can be easily separated from distillation water [23]. The chemical constituents of essential oils are usually consists of readily volatile monoterpenes, sesqueiterpenes, diterpenes and many other nonterpene compounds with different molecular weight and functional group [24]. Essential oils of several plants show their secondary metabolites which are have the crucial abilities as antibacterial, antimicrobial, antioxidant, antifungal and pathogen.
Some of the functional properties are basically attributed to gingerol and shogoal. For example, gingerols inhibited the growth of Mycobacterium avium and Mycobacterium tuberculosis (Z. Kamaliroosta, L. Kamaliroosta).Constituents of essential oil of ginger were reported to be mainly mono- and sesquiterpenes [10]. Whereas, the main pungent constituents of ginger are gingerols, shogaols, paradols, and related compounds, gingerdiones, hexahydro curcumin and the metyl ethers of many of these compounds.
Antibacterial activity
Antibacterial is defined as the potential to destroy bacteria or stop the growth of bacteria. Antimicrobials of plant origin have the numerous of therapeutic potential to heal many infectious diseases with least side effects. Moreover, antibacterial from plant-based are cheaper alternatives to the development of synthetic drugs [25]. The secondary metabolites in plants such as tannins, terpenoids, flavonoids, etc., have been found to have antimicrobial properties [26]. Medicinal aromatic plants and their essences are rich in antibacterial compounds and it is anticipated that phytochemicals with sufficient antibacterial efficacy will be used for the treatment of bacterial infections [27]. Phenols with a hydroxyl group is able to bind with enzymes and inhibit the action of bacteria. Other compounds such as flavonoids, tannins, quinones and coumarins have been extracted and have shown inhibitory effect on a lot of bacteria strains as well as fungi and yeast [28].
The bacterial microorganisms are categorized into Gram-positive bacteria and Gram-negative bacteria. First difference of Grampositive and Gram-negative bacteria is that Gram-positive bacteria retain the crystal violet stain during gram staining, while, Gram-negative bacteria do not retain the crystal violet stain during gram staining. Besides that, Gram-positive bacteria appear in purple color under the microscope, whereas, Gramnegative bacteria appear in pink color under the microscope. Also, outer membrane and peptidoglycan layer is thick and multilayer for Gram-positive bacteria, while, outer membrane is absent and peptidoglycan layer is thin and single-layered for Gram-negative bacteria. Moreover, cell wall of Gram-positive bacteria is smooth and contains non-lipopolysaccharide content, meanwhile, cell wall of Gram-negative bacteria is wavy and contains lipopolysaccharide. Last but not least, lipid and lipoprotein content is less in the cell of Gram-positive bacteria, while, lipid and lipoprotein is a lot in the cell wall of Gramnegative bacteria [25].
Infectious diseases are the leading factor of death worldwide. It kills almost 50,000 people every day [29]. An exponential growth in the incidence of new and re-emerging infectious diseases have urged the action to discover new antimicrobial compounds with diverse chemical structures and novel mechanism of action. To date, drug resistance to human pathogenic bacteria has been generally reported worldwide. However, the scenario is alarming in developing as well as developed countries due to indiscriminate use of antibiotics. Continuation of the emergence of antibiotic resistance and drug resistance pathogenic microbial strains makes it is crucial to discover for further potential antimicrobial compounds.
For example, essential oils of leaf and rhizome of halia bara exhibited better antibacterial activity against the Gram-positive bacteria than against the Gram-negative bacteria. Based on previous study, Gram-negative bacteria showed more resistant to essential oils compared to Gram-positive bacteria [30]. The leaf oil and rhizomes oil of halia bara is different in chemical constituent, the former consists mainly sesquiterpenoids and monoterpenoids in approximately equal amounts, while the latter was dominated by monoterpenoids.
Zingiber spp. produces hundreds to thousands of diverse chemical compounds with different biological activities. Previous studied has been conducted on the leaf and rhizome oils of halia bara which was tested against three Gram-positive (B. licheniformis, B. spizizenii, S. aureus) and three Gram-negative (E. coli, K. pneumoniae, P. stutzeri) bacteria. This study has revealed the bioassays of the leaf oil and oil from the rhizomes of halia bara possessed moderate antibacterial activity against all tested strains, which could have resulted from the presence of caryophyllene oxide, a-pinene, a-terpineol, linalool, 1,8-cineole and geraniol, compounds that are known to possess antibacterial activity. Even though present in low concentrations in the rhizome of halia bara, the aforementioned constituents could have imparted a significant effect on the antibacterial activities of the oils via a synergistic effect [31-33].
Study site
This study was conducted in five locations in Tambunan District which are Rafflesia Forest Reserves (N5.77728, E116.34356), Mahua Waterfall Forest (N5.79684, E116.40846), Trusmadi Forest Reserve (N5.55359, E116.50662), native forest reserve and cultivated local land.
Ethnobotanical survey
Informants were divided into two categories. The first category was the elders and knowledgeable informants such as the practicing medicines men or women. The second category of the informants was common villagers. The number of the informants was depend on the availability or existing of the knowledgeable practicing medicine men or women and common villagers which involved 10 informants. Other villagers which were chosen and asked about their traditional knowledge on the uses of Zingiber species.
Ethnobotanical information was gathered through semistructured interview where there is a form of questions written by the researchers used in face-to-face interactions with the informants. The survey questions were prepared in the fill-in-theblank format, which consisted of questions and blank sections were provided for the interviewer to fill with the given answer by the informants. Questions that were asked during the interview session shown in Figure 1.
Figure 1: Map showing of the three locality study sites (green dots) in Tambunan District.
Another part of the interview, in loco (on site) observation was employed to observe the plants cited and to collect samples for botanical identification. Sampling sites for the specimens were carried out at the nearby forest and at the area around the informant ’ s house. GPS coordinates of the location of the interview session and sampling sites were noted.
Data analysis of ethnobotanical interview
The level of similarity between information provided by different informants was calculated using the Informants ’ Consensus factor, FIC formula (Trotter and Logan, 1986).
The Informants’ Consensus factor, FIC used as shown below,
FIC=Nur-Nt/(Nur-1)
Nur was used to calculate the number of ginger application reports from informants for a particular ginger category while Nt was applied to calculate the number of taxa of species that are used for that plant usage category for all the informants. Values should be in range of 0.00 to 1.00, where “1” was used to indicate the highest level of informant consent.
Collection and identification of fresh plants
Zingiber spp. was collected in the sampling sites for further identification and confirmation of species. Besides that, permission was gained from the Forestry Department Sabah, Tambunan for the plants collection. The collected plants were attached with voucher specimens for identification. Each of the specimens were tagged with a unique number and placed in plastic. The collected specimens were arranged and placed between the newspaper before bringing it to ITBC to be dried and identified.
Plants for phytochemicals and bioassays investigations
After the interviews, the common medicinal gingers of Zingiber spp. were selected for phytochemical analysis. The plant samples were selected based on the availability of the presence of their flowers, fruits, leaves, pits, and rhizomes. Candidate species for investigation of its chemical components shall be available in sufficient amount which was likely to be around 500 g to 1 kg. Ginger samples were obtained at different locations based on the amount and availability of the sample. Most sample was be collected at the Trus Madi Forest Reserve because the Zingiber spp. were mainly found there.
Chemical extraction
Chemical extraction was prepared to obtain the essential oil of the gingers specimen. Rhizome, leaves, and pith of selected Zingiber species that have an aromatic smell undergo essential oil extraction through hydro-distillation. The species that subjected to essential oil extraction is the Zingiber spp. that are available in sufficient amount (500 g to 1 kg).
Extraction of essential oil from Zingiber species by hydrodistillation.
Hydrodistillation was carried out by weighing the fresh Zingiber spp. rhizomes, leaves, and pith. The selected parts of Zingiber species were be cut into 1 cm long pieces, packed inside a hydrodistillation apparatus and submerged in distilled water. Approximately 4 ml of n-pentane was added in the apparatus continuously as solvent. The sample was heated for about 8 hours continuously. After 8 hours, both the distilled water and the n-pentene were collected in separate conical flasks. Sample was kept in the refrigerator until partitioning.
Then, partitioning was done by using a separate funnel and amber tube. The water fraction was partitioned with n-pentane while the n-pentane fraction was partitioned with water in the ration 1:3. The n-pentane later from partition was collected in a clean conical flask and sodium sulphate anhydrous (Na2So4) was added until it glided in the flask. The mixture was left for an hour to remove moisture. Next, the solution was filtered with filter paper and evaporated with rotary evaporator using a round-bottom flask at 3°C, 90 rpm. The essential oil was transferred into a weighed vial and kept at -20°C.
Chemical profiling
Gas Chromatography-Mass spectrometry (GC-MS) was used to study the presence chemical constituents in each of the Zingiber spp. samples.
Gas Chromatography-Mass spectrometry (GC-MS)
Gas Chromatography-Mass Spectrometry was used to analyze the chemical constituents of the essential oil susing a system with capillary column (30 m × 0.25 mm; 0.25 μm film thickness). Helium acted as carries gas with flow rate of 1 Ml min-1. An electron ionization system with energy of 70 ev was applied for detection in GC-MS. The injector temperature was adjusted to 240°C while the mass transfer line set at 220. A total of 1 μl of the sample was transferred into a small vile and dissolved in nhexane before it was injected in the GC-MS using AOC5000 in split mode at a ratio of 1:20 with the total run time of 50 minutes. High purity helium was used as the carrier gas at a constant flow rate of 0.8 ml per minute. The initial temperature was set at 50°C, heated at rate of 3°C per minute to 280°C and held isothermally for 5 minutes. The components of ginger essential oil will be identified on the basis of the comparison of their relative retention time [34]. The volatile compounds will be identified by comparing their mass spectra with standards from the National Institute of Standards and Technology (NIST) 1998 and Shimadzu ’ s Flavours and Fragrance of Natural and Synthetic Compounds (FFNSC) version 1.2. On the other hand, retention indices were determined based on a homologous series of n-alkanes (C8-C40) (Custom Retention Time Index Standard, Restek Corporation, USA).
Antibacterial bioassays
Disc Diffusion Assay was applied in this research to study the potential of extracted essential oils to inhibit bacteria growth.
Types of bacteria used for antibacterial bioassays
The antibacterial activity of the essential oil extracts were determined by agar well diffusion assays. Each essential oil extracts from selected plant parts of Zingiber spp. will be tested against two species of pathogenic bacteria, Escherichia coli (B1) and Staphylococcus aureus (B2). The bacteria were obtained from the Faculty of Science and Natural Resources, UMS (Table 1).
| No | Bacteria | Label |
|---|---|---|
| 1 | Escherichia coli | B1 |
| 2 | Staphylococcus aureus | B2 |
Table 1: List of pathogenic bacteria used in Disc Diffusion.
Types of solvent, positive and negative control used for antibacterial bioassays
Samples from the essential oil are prepared by diluting the essential oil with ethanol, which acted as a solvent. One concentration was prepared for each plant sample, which was 50 μg/mL. A positive and negative control were also prepared to ensure the solvent did not affect the results. The positive control is antibiotic Kanamycin, while the negative control is pure ethanol.
Preparation of nutrient agar and nutrient broth
Firstly, nutrient broth and nutrient agar were prepared and autoclaved to sterilize the medium. Subsequent to autoclaving, unsolidified nutrient agar was poured onto the petri dish. One loopful of each bacteria species were pre-cultured bacterial suspension was seeded on solidified nutrient agar. Secondly, each of the 2 types of bacteria were inoculated and cultured in nutrient broth. This preparation was done in biosafety cabinet. Nutrient broth was then left to incubate for 24 hours to form bacterial colony.
Preparation of sample for Disc Diffusion Assay
On the next day, the 20 μL diluted sample was loaded onto each paper discs (6 mm) and was left to dry under safety cabinet. After that, 100 ml solution from the medium broth containing bacteria was pipetted and streak onto the surface of nutrient. Paper disc containing sample solution was placed firmly onto the nutrient agar by using sterile forceps and parafilmed before incubated at 30°C for 24 hours.
Replication of agar well diffusion method was done based on the availability of sufficient volume (μL) of sample and the best sample which exhibited best inhibition zone was taken for further analyze. After 24 hours, inhibition zone was measured and results were recorded as mean diameter (mm) (Table 2)
| Type of sample | Weight of sample for antibacterial bioassay (μg) | Concentration of sample (μg/mL) | Volume of sample (μL) |
|---|---|---|---|
| Z. pseudopungens inflorescences |
12500 | 50 | 262.5 |
| Z. chlorobracteatum inflorescences |
8000 | 50 | 168 |
| SP3 inflorescences |
2800 | 50 | 58.8 |
| SP1 rhizome |
15600 | 50 | 327.6 |
| SP2 rhizome |
3400 | 50 | 71.4 |
| SP3 rhizome |
22500 | 50 | 472.5 |
| SP1 leaves and stem |
26800 | 50 | 562.8 |
| SP2 leaves and stem |
4300 | 50 | 90.3 |
| SP3 leaves and stem |
2400 | 50 | 50.4 |
Table 2: List of information prepared before antibacterial bioassay was conducted.
Interview sessions had been conducted with the local people in Tambunan District. The interview sessions involved data collection about the significant applications of ginger in their daily basis and the potential of ginger collected used in their medicinal tradition. Ethnobotanical data of ginger applications as valuable plant were taken based on ratio of genders and their frequency of using three types of studied Zingiber spp. in daily basis as shown in Figure 2 below.
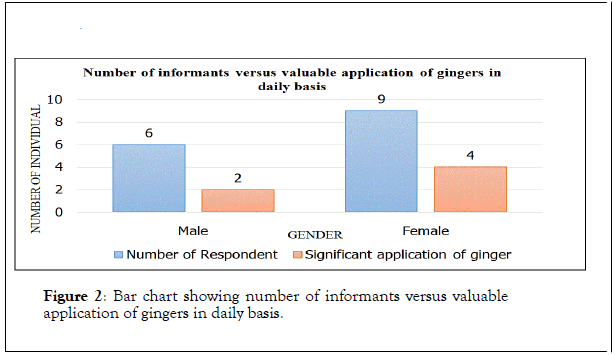
Figure 2. Bar chart showing number of informants versus valuable application of gingers in daily basis.
Table 3 shows the three species of Zingiber and their ethnobotanical uses. All species are indigenous to Sabah and found grow inside undisturbed forests in Tambunan District. All species were found grows in different places and elevations.
| No. | Botanical name (abbreviation) | Vernacular name (s) in Dusun Tambunan | Plant’s part used | Ethnobotanical uses | Locality of collection and habitat |
|---|---|---|---|---|---|
| 1. | Zingiber Z. pseudopungens(SP1) | Teriwad | Whole plants | Ornamentals. | Mahua waterfall, Tambunan. River side, shady area. |
| 2. | Zingiber chlorobracteatum (SP2) | Hayehtahun, Teriwad, Langkuas. | Stem, rhizome, Inflorescence, |
Inhibit poison of venom from snake, can mix with soup, treat cancer, lower blood pressure, ginger tea as blotting remedy, post-maternal herbs, anti-inflammatory medicine | Timbou Village, Tambunan. Forest, expose area |
| 3. | Zingiber coloratum(SP3) | Telidus, Layo | Rhizome, Soft stem |
Stop bleeding of injury, treat diabetes, blotting remedy, able to treat eczema, anti-inflammatory medicine | Rafflesia Forest Reserve, Tambunan. Forest, open areas. |
Table 3: List of Zingiber spp. collected at different locations, plant parts used and the ethnobotanical uses.
Figure 3 below showing the plant’s parts used by the villagers whereby the inflorescence part and rhizome are the most used parts.
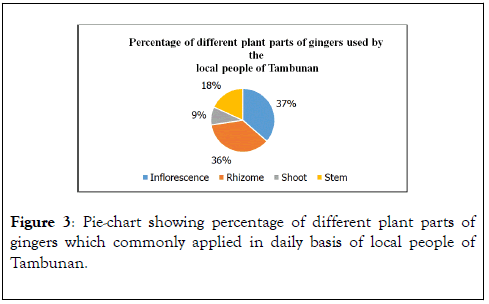
Figure 3. Pie-chart showing percentage of different plant parts of gingers which commonly applied in daily basis of local people of Tambunan.
Figure 4 shows that Zingiber chlorobracteatum is the most edible species by the villagers in Tambunan.
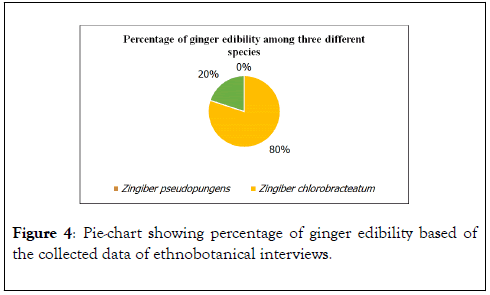
Figure 4. Pie-chart showing percentage of ginger edibility based of the collected data of ethnobotanical interviews.
Informants consensus factor
A statistical analysis was conducted to prove the authenticity of ethnobotanical data from the study sites by using Informant Consensus Factor (FIC). The level of similarity between information provided by different informants was calculated using the Informants consensus factor, FIC formula [35]. The formula used as shown below,
FIC=Nur – Nt/ (Nur – 1)
Nur was used to calculate the number of ginger application reports from informants for a particular ginger category while Nt was applied to calculate the number of taxa of species that are used for that plant usage category for all the informants. Values should be in range of 0.00 to 1.00, where “1” was used to indicate the highest level of informant consent. For example, if few taxa were significant to be used by informants in their daily basis, hence, a high degree of consensus was reached and medicinal tradition is viewed as well-defined [36]. Table 4 shows that miscellaneous uses was the highest percentage, which means vegetables, decoration, flavoring agents, rituals and ornamental have the highest level of informant consent.
| Illness categories (diseases and disorder) |
Number of taxa (Nt) | Number of use reports (Nur) | Informants’ consensus index factor (FIC) |
|---|---|---|---|
| Dermatological disorder (inflammations, eczema, inhibit snake’s venom) | 2 | 3 | 0.5 |
| Gastrointestinal disorder (stomachic) | 2 | 3 | 0.5 |
| General health (cancer, blood purifier, blood coagulant, high blood pressure, headache) | 2 | 5 | 0.75 |
| Miscellaneous (vegetables, decoration, flavouring agents, rituals, ornamental) | 2 | 8 | 0.86 |
Table 4: Statistical analysis showing informant consensus factor (FIC) based as illness categories.
Sample extraction
There are nine types of essential oils which were extracted by using hydrodistillation from three different species. The essentials oils were extracted from their inflorescences, blended rhizome, and leaves blended together with the stem respectively.
Essential oil of Zingiber spp. by hydrodistillation
Each samples of Zingiber spp. of weight ranging from 40 g to 310 g of frozen cut and blended, inflorescences, rhizomes and leaves mixed stems were extracted by using hydrodistillation (Figure 5). Clevenger type to yield grams of essential oils as shown in Table 5 below. The yield percentage of extracted essential oils is calculated by using this formula
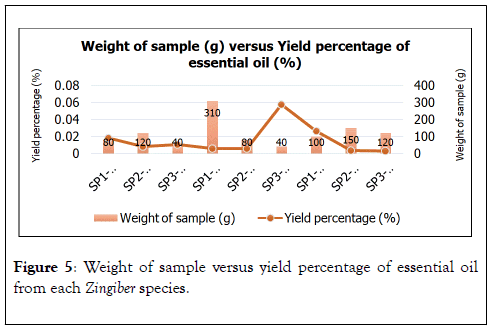
Figure 5. Weight of sample versus yield percentage of essential oil from each Zingiber species.
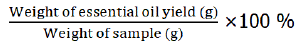
| Type of sample | Weight of sample (g) | Weight of essential oil yield (g) | Yield percentage (%) |
|---|---|---|---|
| SP1-I | 80 | 0.0144 | 0.0180 |
| SP2-I | 120 | 0.01 | 0.0083 |
| SP3-I | 40 | 0.0043 | 0.0108 |
| SP1-R | 310 | 0.0183 | 0.0059 |
| SP2-R | 80 | 0.0048 | 0.0060 |
| SP3-R | 40 | 0.0231 | 0.0578 |
| SP1-LS | 100 | 0.0268 | 0.0268 |
| SP2-LS | 150 | 0.0054 | 0.0036 |
| SP3-LS | 120 | 0.0035 | 0.0029 |
Note: I: Inflorescence; R: Root, LS: Leaves
Table 5: Weight of sample (g), weight of essential oil yield (g) and yield percentage (%) of each samples.
Gas Chromatography–Mass Spectrometry (GCMS)
Tables below show only the top 10 chemical constituents with high concentration percentage (%) presented in 9 samples of Zingiber spp, which encompassed 3 samples of inflorescences, 3 samples of rhizomes and 3 samples of leaves and stems shown in Table 6a-6c.
| Peak no. | Retention time (mm) | Compound | Percentage of concentration (%) |
|---|---|---|---|
| 27 | 16.14 | 2-Isopropyl-5-oxohexanal | 3.66 |
| 43 | 20.41 | (+)-3-Carene, 4-isopropenyl- | 2.68 |
| 49 | 21.47 | Linalyl formate | 2.30 |
| 52 | 21.85 | Alpha.-ylangene | 2.39 |
| 53 | 22.07 | Ethanol | 2.45 |
| 58 | 23.28 | Neobergamate | 2.16 |
| 59 | 23.37 | (R)-lavandulyl acetate | 4.26 |
| 65 | 24.57 | Isoaromadendrene epoxide | 10.61 |
| 75 | 26.27 | Bicyclo-heptane-7-methanol, | 3.43 |
| 93 | 32.52 | Undecanoic acid | 5.79 |
Table 6a: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of inflorescence SP1.
| Peak no. | Retention time (mm) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 9 | 10.52 | 5,10-Dioxatricyclo[7.1.0.0(4,6)]decane | 3.69 |
| 17 | 13.58 | 7-Octen-2-ol, 2-methyl-6-methylene- | 1.28 |
| 24 | 17.31 | Ocim-(4E,6Z)-ene | 1.28 |
| 28 | 18.98 | Cyclohexane, 1,2,4-triethenyl- | 12.33 |
| 40 | 21.59 | Beta.-ylangene | 3.48 |
| 43 | 21.98 | Cyclooctene, 5,6-diethenyl-, | 5.29 |
| 49 | 23.61 | 1,6-Octadien-3-ol, 3,7-dimethyl-, | 1.12 |
| 60 | 26.25 | Bicyclo-hexane-6-methanol, | 5.92 |
| 80 | 32.52 | Undecanoic acid | 8.21 |
| 99 | 38.70 | Hexadecane | 1.88 |
Table 6b: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of inflorescence SP2.
| Peak no. | Retention time (min) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 25 | 19.97 | 1,2,4-Methenoazulene, | 8.04 |
| 30 | 20.97 | Linalyl formate | 26.82 |
| 36 | 21.87 | 1,Z-5,E-7-Dodecatriene | 1.99 |
| 37 | 21.97 | Cyclooctene, 5,6-diethenyl-, cis- | 1.66 |
| 38 | 22.08 | Neral | 3.74 |
| 40 | 22.58 | Bicyclo[2.2.1]heptane, | 3.94 |
| 42 | 23.36 | Oxiranemethanol, 3-methyl-3-(4-methyl-3-penten | 1.12 |
| 49 | 24.74 | Ethanol, 2-(3,3-dimethylcyclohexylidene)-, (Z)- | 1.16 |
| 78 | 32.44 | 9-Oxononanoic acid | 1.10 |
| 92 | 47.18 | Oxalic acid, allyl decyl ester | 1.10 |
Table 6c: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of inflorescence SP3.
Bacteria culture
Both of the E. coli and S. aureus, were added in form of bacterial inoculum into the 100 ml of nutrient broth in falcon conical tubes. After 24 hours, nutrient broths turned cloudy which indicated the presence of bacteria in the broth medium shown in Tables 7a-7c and Tables 8a-8c.
| Peak no. | Retention time (min) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 35 | 23.31 | Neobergamate | 13.85 |
| 40 | 24.64 | Isoaromadendrene epoxide | 52.25 |
| 48 | 26.32 | Bicyclo[4.1.0]heptane-7-methanol, | 43.79 |
| 54 | 29.69 | Trimenal | 11.97 |
| 57 | 32.59 | Undecanoic acid | 76.78 |
| 60 | 35.47 | cis-Z-.alpha.-Bisabolene epoxide | 15.58 |
| 61 | 35.99 | 3-Trifluoroacetoxytetradecane | 11.33 |
| 62 | 36.20 | Longipinanol | 16.91 |
| 64 | 36.77 | Cyclotridecane | 12.58 |
| 66 | 40.55 | Caryophyllene | 14.21 |
Table 7a: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of rhizome SP1.
| Peak no. | Retention time (min) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 42 | 24.33 | Isoaromadendrene epoxide | 8.97 |
| 61 | 29.69 | Trimenal | 20.51 |
| 68 | 32.51 | Undecanoic acid | 10.42 |
| 77 | 35.47 | 2,2,6-Trimethyl-1-(2-methyl-cyclobut-2-enyl)-hepta-4,6-dien-3-one | 10.18 |
| 80 | 36.19 | 2(1H)-Naphthalenone, | 12.32 |
| 89 | 40.37 | Cyclohexanecarboxylic acid, heptyl ester | 18.14 |
| 90 | 40.59 | Caryophyllene | 77.65 |
| 94 | 45.11 | Drimenol | 48.11 |
| 95 | 45.21 | Heptadecane, | 23.47 |
| 96 | 45.65 | Corymbolone | 35.04 |
| 97 | 46.69 | Octadecane | 24.86 |
| 98 | 46.99 | 3,7,11-Tridecatrienoic acid, | 43.47 |
Table 7b: Chemical profiling of the top 12 highest concentration percentage (%) present in compound of rhizome SP2.
| Peak no. | Retention time (min) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 10 | 10.55 | 5,10-Dioxatricyclo[7.1.0.0(4,6)]decane | 3.10 |
| 16 | 16.06 | Borneol, trifluoroacetate (ester) | 0.89 |
| 36 | 20.94 | Linalyl formate | 1.00 |
| 38 | 21.37 | .beta.-copaene | 4.48 |
| 41 | 21.98 | cis-anti-cis-Tricyclo[7.3.0.0(2,6)]-7-dodecene | 2.62 |
| 42 | 22.11 | Cyclooctene, 5,6-diethenyl-, trans- | 1.81 |
| 43 | 22.53 | Cyclohexene, 1,3-diisopropenyl-6-methyl- | 9.42 |
| 47 | 23.39 | Neobergamate | 11.85 |
| 49 | 24.12 | 5,9-Undecadien-1-yne, 6,10-dimethyl- | 0.86 |
| 57 | 25.84 | Dihydrocarveol acetate | 7.13 |
| 58 | 26.21 | Bicyclo[4.1.0]heptane-7-methanol, | 12.62 |
Table 7c: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of rhizome SP3.
| Peak no. | Retention time (min) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 48 | 22.10 | Cyclohexene, 1,3-diisopropenyl-6-methyl- | 11.73 |
| 53 | 23.32 | Neobergamate | 11.01 |
| 60 | 24.66 | Isoaromadendrene epoxide | 48.72 |
| 69 | 26.17 | 2-Bicyclo[3.3.1]non-6-en-3-ylpropan-2-ol | 18.77 |
| 70 | 26.32 | Bicyclo[4.1.0]heptane-7-methanol, 1,5,5-trimethyl-2- | 42.76 |
| 93 | 32.53 | Undecanoic acid | 58.22 |
| 97 | 35.32 | Citronellyl butyrate | 78.49 |
| 100 | 36.76 | Cyclohexanone, 3-(4-hydroxybutyl)-2-methyl- | 21.46 |
| 101 | 37.11 | Cyclohexane, 1,1'-(1-methyl-1,2-ethanediyl)bis- | 41.72 |
| 104 | 40.36 | Cyclohexanecarboxylic acid, heptyl ester | 63.08 |
Table 8a: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of leaves and stem SP1.
| Peak no. | Retention time (min) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 73 | 29.69 | 1H-3a,7-Methanoazulene, | 22.5 |
| 83 | 32.53 | Undecanoic acid | 47.48 |
| 85 | 33.39 | Naphthalene, 1,2,3,4,4a,5,6,8a-octahydro-4a,8-dimeth | 15.48 |
| 86 | 33.39 | 7,8-Epoxy-.alpha.-ionone | 31.38 |
| 92 | 35.32 | Citronellyl butyrate | 14.17 |
| 104 | 40.37 | Cyclohexanecarboxylic acid, | 30.58 |
| 112 | 47.17 | Hexadecane, | 67.02 |
| 113 | 48.13 | Heptadecane, | 40.35 |
| 114 | 49.63 | Nonadecane | 44.15 |
| 115 | 51.36 | 10-Methylnonadecane | 75.44 |
Table 8b: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of leaves and stems SP2.
| Peak no. | Retention time (min) | Compound | Concentration percentage (%) |
|---|---|---|---|
| 24 | 20.94 | Linalyl formate | 22.88 |
| 31 | 22.09 | Neral | 13.98 |
| 36 | 23.38 | (R)-lavandulyl acetate | 5.35 |
| 51 | 27.12 | trans-2-Dodecen-1-ol, heptafluorobutyrate | 12.53 |
| 66 | 31.16 | 3-Tetradecen-5-yne, | 17.02 |
| 69 | 32.52 | Undecanoic acid | 47.78 |
| 76 | 35.34 | Citronellyl butyrate | 55.78 |
| 77 | 36.05 | 5-Pentadecen-7-yne | 30.20 |
| 81 | 40.39 | Cyclohexanecarboxylic acid, | 37.22 |
| 82 | 42.10 | Heptadecane | 10.02 |
Table 8c: Chemical profiling of the top 10 highest concentration percentage (%) present in compound of leaves and stems SP3.
Disc diffusion agar method
Bar-charts below show disc diffusion agar method. This method is used to test the effectiveness of Zingiber spp. to inhibit the bacteria growth by observing the diameter (mm) of inhibition zones shown in Figures 6-8.
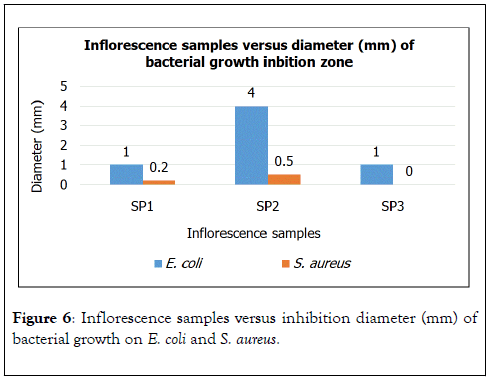
Figure 6. Inflorescence samples versus inhibition diameter (mm) of bacterial growth on E. coli and S. aureus.
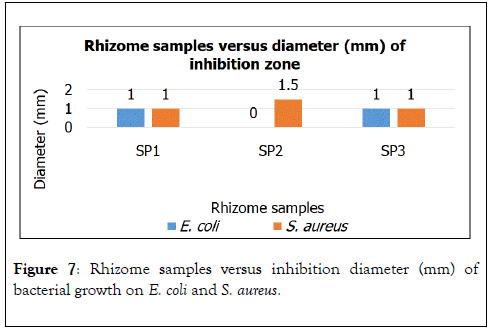
Figure 7. Rhizome samples versus inhibition diameter (mm) of bacterial growth on E. coli and S. aureus.
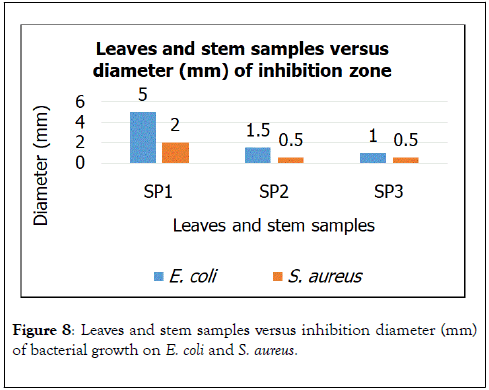
Figure 8. Leaves and stem samples versus inhibition diameter (mm) of bacterial growth on E. coli and S. aureus.
Zingiber Z. pseudopungens, Zingiber chlobracteatum and Zingiber coloratumare the three species of wild gingers collected in various locations in Tambunan District during this studies. Interviews with local people living in Tambunan district, were conducted based on questions. The interview involved 5 males and 9 females by arranging interview sessions with the knowledgeable and practicing traditional medicine men and women. Collecting ethnobotanical data from random local people which were basically the ones selling at vegetables at market was conducted too. Hence, collected data has indicated that female informants show greater number of application of gingers in their daily basis compared to male informants (Figure 2). Analysis of ethnobotanical applications of all Zingiber spp. have indicated that the most frequently used parts are inflorescences of 37%, followed by rhizome with 36%, and shoot with 18%, while, the least used part is stem of 9%. A statistical analysis was conducted to prove the authenticity of ethnobotanical data from the study sites by using informant consensus factor (FIC). Informant consensus of Zingiberaceae plants application ranges from 0.5 to 0.86 per illness category (Table 4). None of the statistic achieved consensus factor of 1.00 which represents moderate level of informant consent towards the value of these Zingiber spp. [36]. The consensus analysis depicted that the miscellaneous category has the greatest FIC of 0.86, general health category has FIC of 0.75, while, dermatological and gastrointestinal category have equal FIC of 0.5. Thus, the FIC of these Zingiber spp. indicates the high degree of consensus was not reached and medicinal tradition is partially well defined [36].
Thus, miscellaneous category shows highest category because most of the informants were afraid of eating the three species Zingiber spp. hence they utilized them only as aesthetic plant such as landscape decoration. General health category shows lower FIC compared to miscellaneous category. This lower FIC of general health category can be supported by information from knowledgeable informant about traditional medicine, it is stated that this ginger especially Zingiber chlobracteatum is not popular to be utilized in traditional medication because there are numerous of other well-known medicinal plants and easy to be found that can be used by them. Both categories of dermatological disorder and gastrointestinal disorder show the lowest and equivalent FIC. This statistical of both categories show several number of the informants utilized gingers specifically as Zingiber chlobracteatum and Zingibercoloratum to treat common health symptoms or problems such as inflammations, eczema and blotting stomach.
Phytochemical studies of all Zingiber spp. samples involved the extraction of essential oils by using hydrodistillation method, chemical profiling by using Gas Chromatography-Mass Spectrometry (GC-MS) and testing process to determine their effectiveness to inhibit bacterial growth of E. coli and S. aureus by using disc diffusion method.
Among all Zingiber spp., inflorescence of Zingiber Z. pseudopungens shows the highest yield percentage of 0.0180%, followed by inflorescence of Zingiber coloratum of 0.0108% and inflorescence of Zingiber chlobracteatum of 0.0083%.
Yield percentages (%) of these inflorescences are not directly proportional to their weight of sample (g). For instance, inflorescence of Z. chlorobracteatum has the greatest weight of sample but produced the least yield percentage as shown in Figure 6. While, inflorescence of Z. coloratum has the least weight of sample but produces higher yield percentage compared to inflorescence of Z. chlorobracteatum as calculated in Table 5.
As for rhizomes, rhizome of Z. coloratum shows that highest yield percentage of essential oil of 0.0578%, followed by rhizome of Z. chlorobracteatum of 0.0060%, while, rhizome of SP3 has yield percentage of 0.0059%. Rhizomes of Zingiber spp. show greater weight of sample but not necessarily produce greater yield percentage of essential oils. For instance, rhizome of Z. Z. pseudopungens and Z. chlorobracteatum produce almost similar yield percentage but their difference in weight of sample (g) are distinct from each other of 310 g and 80 g respectively.
Next, leaves and stems of these Zingiber spp. were mixed together in extraction process. Weight of leaves and stems were weighted equally as one sample which contained both leaves and stems in order to gain a good yield percentage of essential oils. Leaves and stems Z. pseudopungens shows greatest yield percentage of 0.0278%, followed by leaves and stem of Z. chlorobracteatum of 0.0036% and leaves and stem of Z.coloratum of 0.0029%. Regardless of having the least weight of sample among the leaves and stem sample, leaves and stems of Z. pseudopungens produces the highest yield percentage as shown in Figure 6. Meanwhile, leaves and stem of Z.chlorobracteatum and Z. coloratum show a directly proportional relationship between weight of sample and yield percentage of essential oils because their greater weight of sample produces greater yield percentage of essential oils (Table 5).
As displayed in Figure 6, all of the 3 Zingiber spp. show stable yield percentage of essential oils. Thus, as previously studied by Baser and Buchbauer, leaves yield more essential oils compared to inflorescences. In addition, young and mature leaves and inflorescences may significantly affect the oil quality. Commonly, young leaves indicate greater essential oil contents per area unit compared to old leaves. Mostly, oils are produced from flowering plants and thus, highest oil yield is achieved at flowering period.
Based on the Table 5, there is an obvious variation of weight of sample (g) and yield percentage (%) of the extraction Zingiber spp. samples. Taking rhizome of SP3 as an representative, regardless of having one of the smallest weight of sample of 40 g, rhizome of SP3 still managed to produce the greatest yield percentage of 0.0578%. Hence, as revealed, extraction solvents used, extraction methods, extraction temperature, plant extracted part, and extraction period can influence the chemical composition and perhaps their yield percentage of extracted essential oils [37]. Apart from that, seasonal, maturity variation, geographical origin, genetic variation, growth stages, post-harvest such as drying and storage and weight of sample before extraction also affect the chemical composition and may directly or indirectly affect the yield percentage of extracted essential oils [38].
In short, before begin extraction process, the plant sample is ideally to be blended to finer sample by using blender. This preextraction preparation can increase more surface area of the samples to be exposed during extraction process which leads to a better essential oils extraction and thus, produce a better result of chemical composition in chemical profiling. The ideal extracting solvent ration to dry weight sample is 10:1 (volume/ weight) [39].
Gas Chromatography–Mass Spectrometry (GCMS)
In this research, the volatile oils were detected and identified by using Gas Chromatography Mass Spectrometry (GC-MS). This instrumental technique was utilized to identify the chemical constituents present in 9 samples of Zingiber spp. because CG-MS encompasses a gas chromatograph (GC) coupled to a mass spectrometer (MS) which is suitable to be utilized to analyze compounds with multiples of relatively low molecular weight compounds. The complex chemical compound such as essential oils may be separated, identified and quantified in GC-MS.
Also, the identification of chemical compounds in this study is further discussed based on the peak number, retention time and concentration percentage (%) of chemical compounds. It is found that plant parts such as inflorescences, rhizomes, leaves and stems of Zingiber Z. pseudopungens (SP1), Zingiberchlobracteatum (SP2) and Zingibercolaratum (SP3) contained natural volatile oils. These volatile oils can be accumulated on oils cells, secretion ducts or glandular hairs of plants, modified parenchymal cells, resin canals, oil tubes known as vittae, lysigenous cavities, schizogenous passages or gum canals [40]. Moreover, 9 studied samples of Zingiber spp. were identified of containing numerous of chemical compounds such as monoterpene hydrocarbon, sesquiterpene hydrocarbon, diterpene hydrocarbon, oxygenated monoterpene, oxygenated sesquiterpene, hydrocarbon, phenol, alcohol, ketone, acid, ester, ether, amine, and amide.
High concentration percentage of compound shows the major chemical constituents present in the volatile oils. Hence, Tables 6a-6c show the major compound found in 3 inflorescences of Zingiber spp. Compound of isoaromadendrene epoxide were found to be present in inflorescences of SP1, SP2 and SP3. In SP1, the highest concentration of isoaromadendrene epoxide was 10.61%, this compound was found only 0.45% in SP2, while, isoaromadendrene epoxide was found to be least found in SP3 of only 0.31%. Besides that, acid such as undecanoic acid was found major in inflorescence of SP1 with 5.79% and inflorescence of SP2 with 8.21%. While, this compound was absent in inflorescence of SP3. Undecanoic acid is major in both SP1 and SP2 inflorescences because the highest concentration percentage in both samples were only up until 10.61% and 12.33% respectively among the chosen top 10 of highest concentration percentage. Also, cyclohexanecarboxylic acid and heptyl ester are found in inflorescence of SP1 of highest concentration of only 0.39% but absent in inflorescence of SP1 and SP3 as shown in Table 3.
In rhizomes, sesquiterpenoids were dominant. Sesquiterpenes such as β-caryophyllene were present in these three Zingiber spp. For instance, caryophyllene<14-hydroxy-(Z)->8 showed the highest concentration percentage in rhizome SP2 of 77.65% while only 14.21% in rhizome SP1 as shown in Figures 7 and 8. Caryophyllene<14-hydroxy-(Z)->only exhibited 0.44% at peak number 86. Besides that, isoaromadendrene epoxide presented in rhizomes of SP1, SP2 and SP3. As shown in Table 7a Isoaromadendrene epoxide in rhizome SP1 exhibited the greatest concentration percentage among the compounds present of 52.25%, and in rhizome SP2 this compound only exhibited 8.97% as given in Table 7b. The least concentration percentage was in in rhizome SP3 of only 0.20% at peak.
Besides that, neobergamate existed dominantly in rhizome SP1 and rhizome SP3 of concentration 13.85% and 11.85%. While, in rhizome SP3, neobergamate was absented. Moreover, trimenal<Z->existed as major compound in both rhizomes SP1 (11.97%) and SP2 (20.51), whereas, it is absented in rhizome SP3.
In leaves and stems, acid such as cyclohexanecarboxylic acid presented as dominant compound as they exhibited in high concentration in SP1 (63.08%), SP2 (30.58%) and SP3 (37.22%) as given in Tables 8a-8b. Besides that, undecanoic acid also found in all leaves and stem of these 3 species. SP1 contained 58.22%, SP2 contained 30.58% and SP3 contained 37.22% of undecanoic acid. Next, citronellyl butyrate was found high in concentration of 78.49%, 14.17%, 55.78% in leaves and stems of SP1, SP2 and SP3 respectively as shown in Tables 8a-8b. Futhermore, isoaromadendrene epoxide was detected in leaves and stems of these 3 Zingiber spp. SP1 contained 48.72%, SP2 only contained the least concentration percentage of only 1.86% while SP3 contained higher concentration of isoaromadendreneepixode compare to of 2.85% to SP1. On the other hand, diterpene hydrocarbon such as sabinol<trans- >presented as minor compound in the leaves and stems of these 3 ginger species. Sabinol<trans->in SP1 was only 0.17%, SP2 contained 5.36% and SP3 only contained 0.07% which shows SP3 exhibits very little amount of this diterpene.
In short, the variety of accumulation of essential oils different parts of plants and composition of essential oils may be varied on different species of plants and even the exact same species. This phenomenon may happen due to geographical, environmental condition of plant growth, plant organs, predistillation handling, extraction technique and storage. Moreover, the plant maturities and difference chemotype expression also influence the essential oils production. The distinctions in chemical compounds production between plants are crucial to indicate their difference in biological and pharmacological activities through the essential oils analysis [41]. In addition, removal of moisture content is vital to gain good quality peaks in GC-MS analysis. High moisture content influences the extractions, thus limiting the yield of chemical compounds from fully extracted.
Chemical constituents and ethnobotanical applications of Zingiber spp.
Cyclohexanecarboxylic acid and heptyl ester were found in inflorescence of SP1 of highest concentration of only 0.39%. Cyclohexanecarboxylic acid has the potential as antipyretic and anti-inflammatory. Based on ethnobotanical study as shown in Table, none of the informant utilized inflorescence of SP1 as dermatological medicine although its inflorescence contains anti-inflammatory compound.
Besides that, as stated by informant named Nusariah, forest ginger such as SP3 was never been consumed by them because the inflorescence of this ginger has the characteristics of catching insects. This statement can be supported by Sabulalet, because it was identified that volatile compound such as β- caryophyllene is emitted by plants into the atmosphere in response to herbivore attack. Besides that, volatile oils in plant acts as insect repellents because they contain terpenes and phenolic compounds [42].
Based on the collected data of ginger application, some informants utilize Zingiber Z. pseudopungens (SP1) as landscape decoration. SP1 has beautiful pinkish inflorescence grow from the ground. The presence of terpenoids act as intermediates in the biosynthesis of carotenoids to produce color and as attraction to the pollinators such bees, birds, and butterflies [43]. Apart from having colors, the scents of essential oils are strong attractor or repellent for insects. Zingiber spp. commonly an aromatic plants, their fragrance is useful in plant reproduction. For instance, the presence of linalool in flowering plants influences the selective attraction of bees, months and butterflies while repelling flies and fungus by the unpleasant amines such as putrescine [43].
Rhizomes of SP1, SP2 and SP3 may be valuable to be used as dermatological medicine. The availability of β-caryophyllene present in rhizomes of these in gingers gives the potential as anti-inflammatory and anaesthetic drugs. When the essential oils applied on the skin, they are absorbed and transported through connective tissues, lymphatic tissues or circulatory system to reach the target organ [42]. Thus, as shown in Table 3, there are ethnobotanical data collected shows some informant utilized gingers such as SP2 and SP3 as anti-inflammatory medicine.
Besides that, highest in FIC of miscellaneous category as indicated in Table 3, has proven that rhizomes Zingiber spp. especially SP2 is often used as flavoring agents. Utilization of rhizomes of SP2 in cooking could be due to the presence of β- caryophyllene because this compound often found in spice blends and food products.
Disc diffusion agar method
Disc diffusion agar method is used to test the effectiveness of 9 samples from different plant parts of 3 Zingiber spp. to inhibit bacterial growth of Escherichia coli and Staphylococcus aureus. Each paper discs (6 mm) was pipetted with 20 μL of essential oils samples. Kanamycin (50 mg/ml) was used as positive control while absolute ethanol as a negative control.
Positive control and negative control were applied to ensure the validity of antibacterial test. Positive control is crucial to indicate that the bacteria in nutrient agar grow normally and to allow comparison of the effectiveness of samples to inhibit bacterial growth. While, negative control acts as solvent that dissolves the essential oils which does not affect the inhibition activities. Thus, antibacterial test is only valid if positive control shows no growth and negative control shows growth. After 24 hours of incubation, diameter of inhibition zones which showed clear zone around the disc were measured (Ballester-Costa et al., 2013). The inhibition zones in of Zingiber spp. samples were measured in form of diameter (mm).
Based on the diameter of inhibition zones, inflorescences of 3 species samples were found to be able to inhibit bacterial growth of E. coli but only inflorescence of SP3 was unable to inhibit bacterial growth of S. aureus. As given in figure, SP2 shows the greater diameters of inhibition zone on both E. coli and S. aureus. Antibacterial compound such as methyl ester of 0.03% presents in inflorescence of SP2 while absent in SP2 and SP3.
Rhizome samples indicated only SP1 and SP3 inhibited bacterial growth of E. coli as given in Figure 8. Besides that, rhizome samples of all the 3 samples showed their potential to inhibit bacterial growth around the discs. Rhizome SP2 shows the highest inhibition diameter of 1.5 mm while rhizome of SP1 and rhizome SP3 show same inhibition diameter of 1 mm.
Next, all the samples of leaves and stems managed to show bacterial growth inhibition of E. coli and S. aureus. Based on Figure 8, leaves and stems of SP1 shows the highest E. coli growth inhibition of up until 5 mm. Leaves and stems of SP2 and SP3 have similar potential to inhibit E. coli growth. On the other hand, when screening on the potential of leaves and stems to inhibit S. aureus growth was done, SP2 shows the highest inhibition of 2 mm, while, SP2 and SP3 show the same bacterial growth inhibition potentials.
The negative results of some samples to show their antibacterial potential could happen due to several biological factor such symbiosis relationship between plant and bacteria itself. For instance, several essential oils are used to attract symbionts in order to gain sufficient nutrient. There are some plants which are unable to get acquire enough nutrients from the soil. Hence, these plants create a symbiosis relationship with bacteria and fungi in order to aid them to absorb or produce the required nutrient for a better growth. Essential oils may contain allomone chemicals to prevent symbionts by impaired their environment for grow through the roots of other plants [43].
Nevertheless, strong inhibitors in the agar serial dilution test may have low water solubility which gives a poor or even negative result in the agar diffusion test. Therefore, it is complicated to assume that negative result of bacterial inhibition by essential oil does not contain antibacterial constituents. That means, active compound of antibacterial may be simply undetected by using this simple method [44-54]. Thus, disc diffusion method has the benefits of simple test where no complex equipment is needed and low cost. But, the disadvantage of agar disc diffusion method is the test is lack of mechanization or automation of test. Apart from that, the size of inhibition zones is affected by several factors such as volatilization of essential oil, disc sizes, amount of compound given to the disc, absorption of sample by the discs, agar type, pH, volume of agar, and microbial strains tested. In short, this method is suitable to be applied as a pre-test, but the results should not be over-rated [55-60].
In 2003, the World Health Organization reported that around 80% of the society in developing countries use plant based traditional medicines for their well-being needs. The traditional medicine is often being utilized based on theories, beliefs and experiences. Thus, to sum up in regards to this study, proper identification of these Zingiber spp. has a vital role in the utilization of this natural wealth and conservation of biodiversity in the state. The persistent ethnobotanical knowledge of these gingers is proof to the essential and valuable nature of their importance in human health. Besides that, ethnobotanical knowledge tends toward disappearance because transmission of this type of information from generation to generation are often only through oral practices without any reliable written document. Hence, documentation of this kind of traditional knowledge can help to secure several information and pave the direction for strengthening ethnobotanical knowledge and socioeconomic upliftment. Besides that, documentation of plants uses can create incentives for future phytochemical and pharmaceutical research to discover many novel drugs.
Volatile compounds of the Zingiber Z. pseudopungens(SP1), Zingiber chlorobracteatum (SP2) and Zingiber coloratum(SP3) were extracted by hydrodistillation method and analyzed by GC-MS. A lot of pharmaceutical industry has been supported by chromatographic method. This instrumental technique is useful in order to perform chemical profiling with specific medicinal properties. In this study, the preliminary phytochemical screening in both polar extract of ethanol and non-polar extract of n-hexane showed the presence of major secondary metabolites in these 3 types of studied Zingiber spp. GC-MS profiling gave the composition of few major compounds. Moreover, antibacterial assays of these oils had demonstrated that the Gram-positive bacteria in this study were more sensitive to the Zingiber spp. samples compared to the Gram-negative bacteria. Further studies are required to discover the mechanisms of biological activities of chemical compounds and bacterial strains and identification of biologically active compounds responsible for the action. In such way, the extracts these 3 studied Zingiber spp. could potentially be a new source of natural drug which may be subjected to food, cosmetics and pharmaceutical industries.
The authors would like to acknowledge the contributions of various persons whom involved in this studies, namely to Assoc. Prof. Dr. Monica Suleiman, the Director of Institute for Tropical Biology and Conservation, Universiti Malaysia Sabah for the financial support and approval of this research studies. This studies would not be accomplished without the full supports from the District Office of Tambunan and various villagers in Tambunan District namely Mr. Medi Kusop (Village Head), Mr. Philip KulipGidam, Mr. Ferdinand Opot George Killoh, Mr. Damian George Killoh and Mr. Justin Gambun from Timbou Village and Mr. JamainAgang of Sunsuron Village. Specimens comparisons were conducted at Sabah Park Herbarium in Kinabalu National Park, Kundasang District and at Sabah Museum Ethnobotanical Herbarium in Kota Kinabalu. Last but not least to Sabah Biodiversity Council office for the approval of research permit.
Citation: Kulip J, Nawan CST, Vairappan CS, Jaumin B (2020) Ethnobotanical and Phytochemical Studies on Indigenous Zingiber spp. (Zingiberaceae) from Tambunan District, Sabah, Borneo, Malaysia. Nat Prod Chem Res. 8:374. DOI: 10.35248/2329-6836.20.8.374
Received: 09-Jul-2020 Published: 30-Jul-2020
Copyright: © 2020 Kulip J, et al. This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.